The centrifugation method separates different components or the suspended particles from the homogenous solution through centrifugal force. A natural gravitational force can also concentrate particles but in an indefinite time.
To increase particle separation efficiency, a centrifugal force function to separate particles by sedimentation property. It separates particles based on their physical properties like size, shape, density etc. and chemical properties like molecular weight and viscosity.
The rate of sedimentation increases and decreases as per the increase in gravitational force and viscosity of the medium, respectively. In this context, we will study the definition, principle, types and uses of the centrifugation technique.
Content: Centrifugation Method
Definition of Centrifugation
The centrifugation method refers to concentrating suspended particles from the aqueous medium, after which the particles segregate by forming discrete bands and pellets, depending on their size and density. A centrifuge apparatus aptly causes the partition of two definite phases, namely supernatant aqueous phase and suspended solid phase.
According to the sedimentation rate, a rotor inside the centrifuge generates a central or centrifugal force that facilitates the separation of a solid and aqueous phase. Based on the particle separation, there are two significant types of centrifugation methods:
- Preparative centrifugation: It is a technique concentrating one component from the other.
- Analytical centrifugation: It is a method that measures the physical, chemical and hydrodynamic properties of the suspended molecules.
Principle of Centrifugation Method
A centrifugation method depends on the gravitational movement of suspended particles, which cause the deposition of high-density particles towards the bottom. It is important to note that higher is the difference between the densities of particles; easy will be the separation.
If the density difference is low, the separation becomes tough, and the gravitational force must be accelerated in such a case. Therefore, a centrifuge accelerates the sedimentation rate by increasing the relative centrifugal force (RCF) measured by the rotations per minute (rpm). If RCF is equal to 100X g, then it is equivalent to the centrifugal force that is a hundred times greater than the force of gravity.
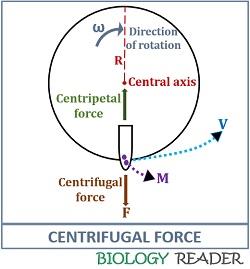
Centrifugal field
A centrifugal force is a kind of force that causes circular motion of the particles and given by an equation:
Centrifugal force = mω2r
Where, m= Mass of particle
ω= Angular velocity in radians per second
r= Radius of a particle from the axis of rotation
ω2r= Centrifugal acceleration
In terms of gravitation force, the equation becomes,
Gravitational force (G)= mg
Where g= Gravitational constant
Interacting Forces
There are some other interacting forces, namely buoyant and frictional force, other than the centrifugal force.
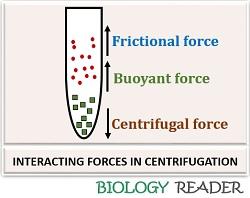
Buoyant force
A buoyant force is an exerted force causing the movement of particles upwards, and it develops as a result of force applied to the particles by the fluid. The opposing buoyant force is smaller for the dense particles.
Buoyant force (Fb)= ρgV
Where ρ= Fluid density
g= Gravitational or centrifugal acceleration
V= Volume of fluid displaced
Frictional force
A frictional force is a kind of force that restricts the movement of dispersed particles in the solution. The frictional coefficient is smaller for the minute particles than the dense particles.
Frictional force (Ff )= fv
Where, f= Frictional coefficient
v= Sedimentation velocity
The net force acting on the particles is equated as,
Net force= Centrifugal force – Buoyant force
= mω2r – ω2rρV (ω2r= Centrifugal acceleration, thus g= ω2r)
In a steady state, the frictional force is equal to the net force.
fv= mω2r – ω2rVρ
= mω2r – ω2rmv̅ρ
= mω2r (1– v̅ρ)
The mass of 1 mole of particles is represented by,
M=mN (Where N is the Avogadro’s number)
fv= Mω2r (1– v̅ρ)/N
Now, the sedimentation coefficient (S) refers to the ratio of velocity to gravitational acceleration.
S= v/ω2r
= M/N. (1– v̅ρ)/f
A sedimentation coefficient is measured by the units of a second or the Svedberg unit (1S= 10-13second).
Types of Centrifugation Method
There are commonly three kinds of centrifugation techniques:
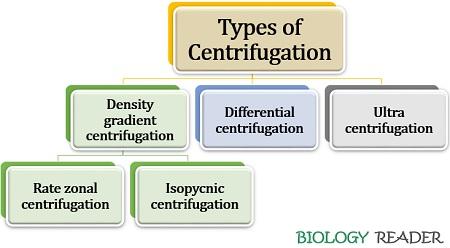
Density gradient Centrifugation
It is another centrifugation method in which the sample containing different molecules mixes with the high-density solution. Density gradient centrifugation makes the use of substrates (sucrose, glycerol etc.) to prepare a solution for the development of a density gradient.
For separating different concentrates, the solution (where the sample has to be mixed) should have low concentration and high diffusing property. Before centrifugation of the sample solution, it should be stored in the uniform mixture for some time.
On centrifugation, the different molecules of different density will occupy the place in a density gradient, respective to their masses. Finally, a concentrated layer of the particles is isolated by puncturing the centrifuge tube.
- Rate zonal centrifugation: In this kind, a sample is centrifuged in a pre–established density gradient.
- Isopycnic centrifugation: Here, a self-generating gradient forms during the centrifugation of the sample.
Rate zonal Centrifugation
It refers to the velocity centrifugation, where the separation of the particles occurs based on their sedimentation rate that directly depends upon the particle mass or size. The particles’ shape also influences the rate of sedimentation.
It makes the use of a high-density sucrose solution of 5-20% to create a density gradient. The sedimentation of dense particles occurs at the bottom of the centrifugal tube, and the light particles remain at the top. Therefore, zonal centrifugation develops discrete bands or zones, respective to the density of the particles present in the sample.
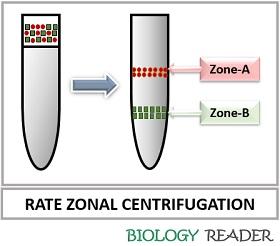
For the separation of bands, a centrifuge tube is prickled from the bottom. The sedimentation coefficient and the mass of the molecule of interest can be determined by this technique. The particle size or mass may affect sedimentation efficiency, as the sedimentation coefficient is proportional to the mass of a particle.
Isopycnic Centrifugation
It refers to the equilibrium density gradient centrifugation where the particles are concentrated based on their buoyant density-independent of particle size and shape. The time taken for the centrifugation of particles does not affect the rate of sedimentation.
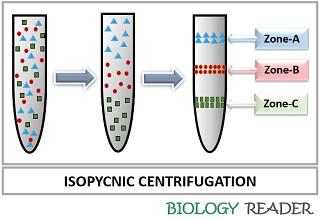
This method is employed for concentrating such particles possessing the same size but different density. In isopycnic centrifugation, 20-70% sucrose solution develops a gradient to separate the different molecules. The sedimentation of biomolecules will not occur until the buoyant density does not equal the density of the gradient.
Differential Centrifugation
It is a centrifugation method that depends upon the differences in the sedimentation rate that directly influences the separation of different components according to their relative mass, shape and density.
Generally, it separates the subcellular components. In differential centrifugation, blend the cells. Pour the ruptured cell components into a centrifuge tube that preserves the integrity of the cell components.
Then, the suspended components in a solution are subjected to initial centrifugation at low centrifugal force for a defined time. The initial centrifugation causes sedimentation of larger molecules at the bottom (as the pellets) and supernatant at the top.
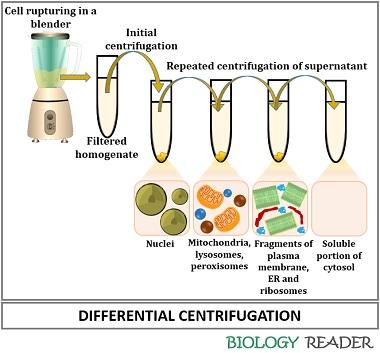
After centrifugation under a high centrifugal force, the supernatant is repeatedly decanted to sediment different cellular components. Therefore, the centrifugal force plus the centrifugal time increases successively after each step.
Ultracentrifugation
This method increases the earth’s gravitational force by using centrifugal force (1,000,000 times) greater than the gravitational force. Ultracentrifugation causes sedimentation of particles as small as 10kDa.
In a homogenous mixture, the dense particles will appear as the pellets at the solvent sink, and the lighter particles will appear as supernatant at the solvent top. Ultracentrifugation uses a dense solution of sucrose or caesium chloride to concentrate particles.
Uses of Centrifugation
Following are the applications of the centrifugation technique:
- Environmental science: In the treatment of wastewater.
- Molecular science: Helps in the extraction of biomolecules like DNA, RNA, protein etc.
- Medical research: Helps in the separation of different components from urine, blood serum etc.
- Chemical science: Helps in the process of uranium enrichment.
- Food science: Helps in the production of skimmed milk by removing fat.
Therefore, we can conclude that the centrifugation technique has broad applicability in research biochemistry, molecular biology, cellular biology and medical science.