The production of streptomycin requires the stock culture of Streptomyces griseus, proper nutrient medium and the other growth-promoting substances. Streptomycin is an antibiotic that shows a broad spectrum against the gram-negative bacteria and is the first antibiotic to cure TB. In 19th October 1943, a scientist Albert Schatz from the Rutgers University was the first to isolate “Streptomycin”.
Albert Schatz was the PhD student, working under the guidance of Selman Abraham Waksman. Along with streptomycin, Waksman and his interns have also discovered several other antibiotics like actinomycin, neomycin, clavacin etc. Streptomycin was the first medicine discovered for the treatment of tuberculosis, which was found to be effective.
According to the World Health Organisation (WHO), streptomycin is the safest medicine for treating tuberculosis. Streptomycin has been included in the list of essential medicine needed for the health system by the WHO. In the year 1952, Waksman won Nobel Prize in physiology and medicine for the discovery of streptomycin.
Content: Production of Streptomycin
Definition
Production of streptomycin can be defined as the process that produces an antibiotic, i.e. streptomycin, using the strain of actinomycetes. Streptococcus griseus is used in the large scale production of streptomycin, which utilizes carbon, nitrogen, mineral source etc. incorporated in the fermentation medium.
Streptomycin: Streptomycin refers to the broad spectrum aminoglycoside antibiotic, which works actively against the gram-negative bacteria. Streptomycin is commercially available as “Hypochloride” with a chemical formula C21H39N7O12.3HCL.
There are three sugars with carbon, nitrogen, oxygen and hydrogen elements in the structure of streptomycin. Some of the physical and chemical properties of the streptomycin are given below in the table.
| Physical and Chemical properties | Streptomycin |
|---|---|
| Physical state | Solid |
| Form | Hygroscopic powder |
| Odour | Faint amine like odour |
| Taste | Slightly bitter |
| Solubility | Soluble in water |
| Nature | Hygroscopic |
| Chemical names | Agrimycin, strepcen etc. |
| Molecular weight | 581.58 g/mol |
| pH | Polar organic base |
| On decomposition | Emit fumes of nitrogen oxide |
Organism Required for Streptomycin Production
Streptococcus griseus is an actinomycete, which is used for the preparation of streptomycin. Krinsky was the first scientist isolated S.griseus during the time of 1st world war from the Russian soil.
S.griseus is commonly present in the soil, and it is gram-negative bacteria. It produces grey mycelium during sporulation and produces grey yellow reverse pigment when it grows in the form colony.
Streptococcus griseus secretes toxin valinomycin, which can cause apoptosis in natural killer cells and mitochondrial swelling in peripheral lymphocytes. Thus, due to the presence of valinomycin, the excessive use of streptomycin can cause neurotoxicity like hearing loss, dizziness etc.
Classification:
- Domain: Bacteria
- Phylum: Actinobacteria
- Order: Actinomycetales
- Family: Streptomycetaceae
- Genus: Streptomyces
- Species: S.griseus
Production Process of Streptomycin
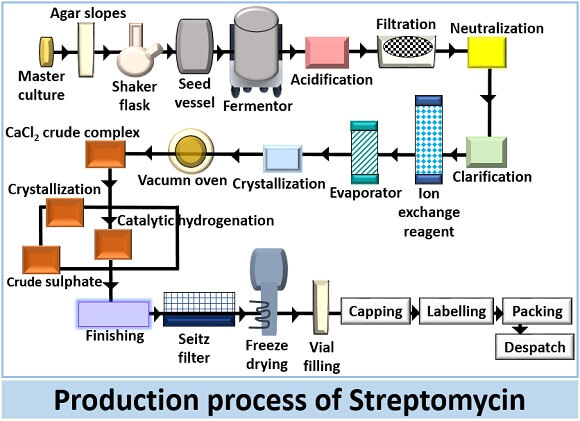
Production of streptomycin is a complex process, which goes through a series of operations under the sterile conditions. Let us summarize the steps in the production process of streptomycin.
Growth of Streptomyces griseus
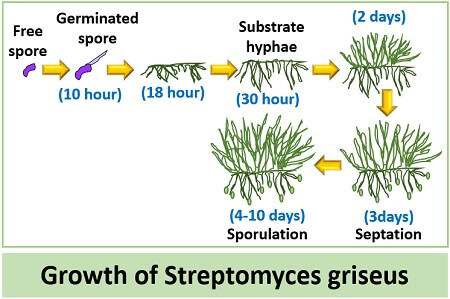
Maintain the hymns of Streptomyces griseus spores in the stock soil culture. Then, inoculate the inoculum of S.griseus into the agar media at 28 degrees Celsius.
After that, the spores of S.griseus sporulate and build up the mycelial network in the flask or seed tanks. The sporulation of S.griseus occurs well in a liquid culture medium, and it forms endospores in low nutrient availability. The growth of S.griseus requires a pH range of 5-11.
Preparation of the Medium
The production of streptomycin requires Hockenhull nutrient medium with the following media composition:
- Glucose: 2.5%
- Soy meal: 4%
- Sodium chloride: 0.25%
- Distillers dried soluble: 0.5%
- pH: 7.6-8.0
For the production of the medium, there must be energy sources like carbon, nitrogen etc.
- Carbon source: Monosaccharide like glucose provides the best carbon source that helps in building up of streptomycin. Sugars like fructose, maltose, starch etc. can also be used as the carbon source, but oligosaccharides and polysaccharides give low yield.
- Nitrogen source: Contents like soy meal, meat extract, ammonium salts etc. serve as the nitrogen source.
- Mineral source: Magnesium, calcium, potassium etc. provides a mineral source and sulphates, phosphates and chlorides.
- Growth stimulating source: Sources like L-naphthalene acetic acid and phenylacetic acid functions as the growth-stimulating factor in the production of streptomycin.
Besides the above sources, compounds like proline, oils, fatty acids, antioxidants are also used to accelerate the streptomycin production. The precursors are not required for the production of streptomycin.
Fermentation
Sterilize all the above contents and fed into the stirred tank fermentor. Then, transfer the bacterial inoculum of about 4-5% to the medium. Maintain a temperature between the range of 25-30 degrees Celsius and pH between 7.6-8 inside the fermentor. The production of streptomycin requires continuous fermentation, and the growth of S.griseus requires continuous aeration and agitation. The fermentation process takes 4-10 days. The fermentation process involves three consecutive phases.
First phase
During the first phase, the S.griseus grows by utilizing soya meal and releases ammonia through proteolytic activity. The pH rises during this phase. S.griseus slowly utilizes the carbon source present in the soy meal and the glucose and thereby producing streptomycin at a slower rate.
Second phase
After the utilization of soy meal and glucose, the ammonia is secreted, and the streptomycin is synthesized rapidly. In this phase, the incubation period lasts for 24 hours to 6-7 days. The pH during this phase remains constant between 7.6 and 8. There is little or no production of mycelium.
Third phase
It is the final phase of fermentation, where the concentration of glucose depletes from the medium that results in cessation of streptomycin production, and the bacterial cells begin to lyse. As a result, the medium’s pH increases by the deposition of ammonia after the cell lysis. Before cell lysis, harvest the fermentative medium for the process of recovery and purification.
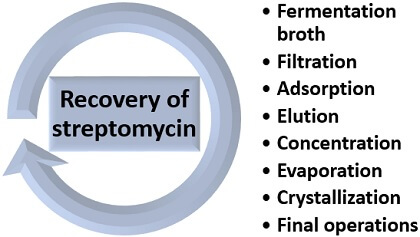
Recovery of Streptomycin
The recovery of streptomycin includes the following stages:
- Filtration: In the recovery of streptomycin, separate the bacterial biomass from the fermentative broth through Oliver’s pre-coated pressure filter. After filtration, the remaining liquid is first acidified, filtered and then neutralized.
- Adsorption: Mix the activated carbon with the clear broth obtained after filtration in a series of three adsorption tanks. Separate streptomycin from the broth to remove impurities.
- Then, the broth is fed to the pressure filter, which releases out the spent broth. Wash the absorbate with dilute alcohol to remove impurities.
- Elution: Elute the streptomycin via ion exchange method by adding dilute hydrochloric acid. In this process, the impurities like metallic salts are removed from the solution, which can contaminate the streptomycin.
- Concentration: Concentrate the remaining solution at 60 degrees Celsius under vacumn by the series of three single-phase evaporators.
- Crystallization: Firstly, dissolve the streptomycin in methanol and then filter it again. Then, treat the filtrate with acetone to obtain a precipitate. Rewash the precipitate with the acetone and later dry it under vacumn.
Final operations
It includes the following steps like:
- Then, dissolve the crystalline streptomycin in the solution containing pyrogen-free water.
- Pass the solution through the special biological filter like Seitz filter to eliminate the microorganisms that can contaminate the streptomycin.
- Then, dry the filtrate under vacumn to form a dried cake.
- Crush the dried cake into a powdered form by employing high vacumn sublimation using sterile steel balls.
- After that, weigh the powdered streptomycin into sterile vials. The whole process needs to be carried out in the air-conditioned room, which provides filtered or stale air through the ultraviolet lamps.
- Finally, package the powdered streptomycin in the rooms having 10% relative humidity.
To avoid or reduce the chances of contamination, you can use special microbial filters, triethylene glycol as a disinfecting agent, and UV lamps. All these factors maintain sterile conditions throughout the production process of streptomycin.
Uses of Streptomycin
Clinical science:
- Streptomycin is very effective against gram-negative bacteria, thus treating the infections and diseases caused by gram-negative bacteria.
- It is widely used in the treatment of severe disease like tuberculosis.
- Streptomycin is used in conjugation with antimycobacterial drugs, to treat pulmonary infections caused by the Mycobacterium avium complex.
- It works as an alternative of Penicillin G used to treat the rat-bite fever caused by Streptobacillus moniliformis for those who are allergic or sensitive to the penicillin.
- Products like streptomycin-B and dihydroxystreptomycin can be obtained from the streptomycin.
- Patients suffering from endocarditis caused by Enterococcus can be treated with streptomycin if they are sensitive to the gentamicin. Tularemia infections, Plague, Brucellosis etc. can also be treated with the streptomycin.
Agriculture science: Streptomycin is used as “Pesticide” that inhibits the growth of bacteria, fungi, algae etc.
Cell culturing: It is used at the time of cell culture, to prevent undesired microorganisms’ growth.
Biochemistry: Streptomycin sulphate is also used in the purification of biomolecules like protein and sometimes nucleic acid.
I am impressed by the construct of your article. I’ve been looking for a good article. I am stumbled on your website, and it brought my search to an end.
It is really helpful.