Synchronous culture is a technique of growing microorganisms at the same stage of their growth cycle under controlled conditions. Forced or mechanical selection methods induce synchrony in the microbial culture.
Forced techniques induce synchrony in microorganisms by the shock treatment (temperature variation) or chemical treatment (nutritional difference). In contrast, the mechanical method uses filtration or centrifugation techniques to collect cells of the same age and size.
The microbial population in synchronous culture never lasts more than 4-5 generations, as the generation time for individuals dividing cells varies considerably.
In this post, we will discuss the definition, purpose, and efficiency of synchronous culture. You will also get to know the key points, mechanical method and induction method of the synchronous culture.
Contents: Synchronous Culture
Definition of Synchronous Culture
Synchronous culture refers to the growth process of the microbial population, where individual cells show synchrony with the other cells in the same culture medium by growing at the same growth phase for the given generation time.
The main characteristic of synchronous growth is that all the microbial cells are physiologically identical by growing at the same division cycle and same generation time. Therefore, we can say that the entire microbial population remains uniform concerning cell growth and division.
Purpose
- By using synchronous culture, we can get the idea of the entire cell crop in the particular stage of their life cycle and their interrelations.
- The measurement of microbial growth in synchronous culture is more accessible than the other growth culture techniques, as the results made on such mass culture are analogous to the measurements made on a single bacterial cell.
- In the synchronous culture, we can elucidate the growth behaviour of the bacterial cells in the same stage of growth.
Key Points
- The entire microbial population tends to show synchrony by altering the culture media’s physical conditions and chemical constituents.
- It is a kind of open cultivation system.
- In synchronous culture, the microbial cells are physiologically similar.
- All the microbial cells in synchronous culture grow at the same generation time and the same divisional cycle.
The Efficiency of Synchronous Culture
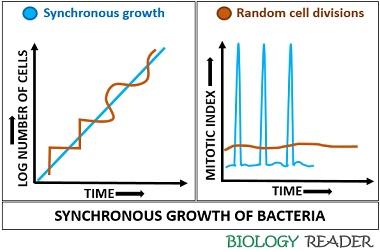
The bacterial cells show asynchronous growth in the random culture medium. However, the microbial cells show synchrony through the selection and induction methods. We could determine the efficiency of synchronous growth by comparing the following two parameters:
- The mitotic index
- Generation time for doubling the microbial population
Let us see the growth pattern of cells by plotting a graph between doubling time vs a logarithmic number of cells and time vs corresponding mitotic indices for a synchronous and random growth, respectively.
Selection of Microorganisms
To select microorganisms and keep them in synchrony, there have been two approaches employed based upon the mechanical and induction selection methods.
Selection by Mechanical Method
It refers to a physical separation method, in which the selection of a synchronous population is carried out according to age and size.
To perform this method, you need to filter the microbial cells to separate the metabolically active small and young cells. A filter retains large cells that are ready to split.
In this way, the large cells are collected from the filter to obtain a synchronous growth by a standard technique known as the “Helmstetter Cumming technique”.
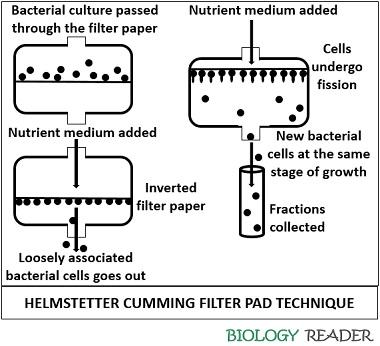
In this method, You need to pass the entire cell population through a filter whose particle size is small, sufficient to trap bacteria. This method uses a cellulose nitrate membrane filter.
Then, invert the filter paper and pass the fresh nutrient medium over it. By doing this, the loosely associated bacteria will wash away through the filter. The large-sized bacteria will remain on the filter paper, and they tend to divide.
Then, collect the sample of this stream, which contains all the newly formed and synchronously dividing cells. The method has one limitation that the population size is petite.
Nowadays, density gradient centrifugation is also used instead of filtration to select microbial cells of the same size and density that can divide at the same stage of their life cycle.
Selection by Induction Method
The other method, i.e. shock treatment, induce cell synchrony at altering temperature, starvation, light exposure, lethal doses of radiation etc.
All these factors can maintain synchrony in the cell population for several generations. Let us discuss some of the factors that can induce synchrony in the cell culture.
Temperature variation: It is the most common factor that induces cell maturation to the same point of fission.
We could observe the change in the culture medium by growing the microbial culture under 37 degrees Celsius and later subjecting the culture medium to 20 degrees Celsius for about 30 minutes.
During this interval, the cells go through maturation to undergo cell division. At 20 degrees Celsius temperature, no bacteria will undergo fission.
But, if you transfer the culture at a temperature of 37 degrees Celsius, all the cells start dividing synchronously. Therefore, the repeated temperature variation can maintain synchrony for a few generations.
Alternations in the media composition: Other than shock treatment, synchrony can also be induced by changing the media composition of the culture medium.
The microorganisms grown in the culture medium deficient or containing the essential growth factor may either promote or inhibit the cell division, which eventually withheld the fission.
Let us suppose the bacterial cells are grown in a culture medium lacking thymine (an essential element). As a result, the process of fission halts for some time. However, the bacterial cells will divide once you transfer the cells to a complete nutrient medium.
Similarly, colchicine is a growth factor inhibiting cell division in culture medium for a certain period. But the effect can be reversed once you transfer the cells to the culture medium free of colchicine.
Conclusion
Therefore, we can conclude that the synchronous culture is of two kinds, namely induction and selection synchrony. The induction synchrony induces the bacterial cell population to undergo fission synchronously via physical or chemical treatment.
Oppositely, the selection synchrony selects cells at a particular stage of the cycle, and later the fractions of bacterial cells grow through their natural cycle.