Flagella staining is a technique examining the presence and arrangement of bacterial flagella under the microscope. It is a specialized staining method, which requires a combination of special reagents to stain the bacterial flagella.
The flagella appear as narrow appendages, which cannot be visualized by employing common stains. Therefore, the flagella staining uses mordants to thicken the bacterial flagella that are too thin to be stained.
Then, the flagella are stained by using special flagella stains and finally examined through bright field microscopy. The flagella staining uses two methods, namely, wet-mount staining or Ryu method and dried-smear method or Leifson’s staining technique.
This post describes the definition and two commonly practised techniques of the flagella staining along with their result interpretations. Also, the meaning and characteristics of the bacterial flagella have been explained.
Content: Flagella Staining
Flagella Staining Definition
Flagella staining refers to one of the specialized staining techniques, which uses mordant and special stains to visualize the flagella. It is quite a complicated method, which requires specific considerations, proper handling and operation, due to the fragility of the bacterial flagella.
The purpose of flagella staining is to check whether a bacteria is motile or non-motile. This special staining procedure helps in the genus and species differentiation based on the bacterial flagella’s shape, size, location, and arrangement.
Bacterial Flagella Meaning
Bacterial flagella are much thinner and simpler in structure. They exist as thread-like and twisted appendages, which span the bacterial cell membrane and protrude out to give motility to the bacteria.
Characteristics
- Size: The length varies from 20 µ to 200 µ.
- Existence: Flagella mainly exist in rod-shaped bacteria and rarely occur in few cocci.
- Localization: They are generally localized as polar or lateral flagella, depending upon the different bacterial species.
- Structure: The bacterial flagella comprise three components, namely a basal body, hook and filament.
- The basal body connects the flagella to the cell membrane and cell wall.
- A hook is generally short.
- The filament appears as a whip-like structure that is several times longer than the bacterial cell. It possesses flagellin protein.
Flagella Staining Techniques
Wet mount and Leifson’s staining method are the two prevalently used techniques of the flagella staining procedure.
Wet Mount Method
It is a more successful and easy method to practice for routine use. The wet mount method is also called the “Ryu method” because it uses Ryu flagella stain to examine the arrangement and number of flagella.
It is hard to study the existence of flagella or identify the species of motile bacteria because of the flagella’s thin size. Therefore, a mordant is used in the staining procedures to visualize flagella, which sticks in the layers of flagella.
Preparation of Ryu stain: It involves the preparation of two solutions (Solution-I and II).
- Solution-I includes the following components in a defined amount:
- 5% aqueous solution of phenol: 10 ml
- Tannic acid: 2 g
- An aqueous solution of aluminium potassium sulphate-12 hydrate: 10 ml
- Solution-II contains a saturated ethanoic solution of crystal violet, in which 12 g of crystal violet is mixed in 100 ml of 95% ethanol.
The final stain is prepared by mixing solution-I and II in a ratio of 10:1. Then, separate the coarse precipitated particles from the stain via filtration using filter papers, and finally store the reagent at room temperature.
Procedure of Wet Mount Technique
- The organism needs to grow at room temperature in a blood agar medium for 16 to 24 hours.
- Put a drop of saline onto the microscope slide.
- Now take a sterilized inoculating loop and remove little inoculum from the culture plate by only touching the margin.
- Then, add the inoculum into the drop of water placed on a glass slide.
- After that, leave a glass slide undisturbed for about 15-20 minutes.
- Afterwards, place the coverslip to the faintly turbid drop of water and immediately view it under the 40-50X of the objective lens.
- If the motile cells are visible, the process is followed by staining the bacterial culture. Add a drop of Ryu flagella stain towards the one edge of the coverslip, which ultimately penetrates the bacterial suspension through capillary action.
- Then, observe the glass slide after 10 minutes, under the light microscope upto the power of 100X.
- Finally, note down the results by examining the presence, number and arrangement of the flagella.
Leifson’s Staining Method
As flagella are too thin hair-like structures, so it’s not possible to visualize them under the microscope. It uses Leifson dye as a special reagent to colour the thin flagella.
We can prepare Leifson’s reagent by simply mixing a mordant (tannic acid) and stain (basic fuschin formed in an alcohol base).
Once Leifson’s stain incorporates within the cell, the tannic acid available gers attach to flagella, and the available alcohol evaporates.
After evaporation of alcohol, the flagella thickness is increased due to the deposition of tannic acid. After the above treatment, we need to treat the cells with the methylene blue reagent, which eventually stains the cell.
Preparation of Leifson’s Stain: To prepare leifson stain, we require the following contents in the given amount:
- Ammonium or potassium alum, saturated water solution: 20 ml
- The tannic acid in 20% water solution: 10 ml
- Distilled water: 10 ml
- 95% of ethyl alcohol: 15 ml
- A saturated ethyl alcohol solution of basic fuschin: 3 ml
Procedure of Leifson’s Staining Technique
- Firstly, take flagellated cell culture slant and put two to three droplets of distilled water into the culture slant dropwise by using a sterile pipette without disturbing the cell growth.
- Now incubate the slant for 20 minutes after adding water into it.
- Afterwards, take one drop from the above-prepared suspension and put it on a clean slide. Then keep a slide in an inclined position.
- The drop needs to flow from one end to another end of the slide to restrict the flagella folding on the cell.
- Now allow the smear to air dry.
- After the liquid completely evaporates, flood a glass slide with Leifson’s stain until you observe a shiny thin film.
- Then wash the slide gently with water.
- Afterwards, treat a glass slide with 1 % methylene blue for one minute.
- Observe the glass slide by putting a drop of oil immersion after washing the slide with water and air-drying.
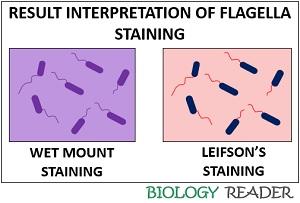
Result Interpretation
- Wet mount staining method: It stains the flagella purple.
- Leifson’s staining method: It stains the flagella red and the bacterial cells blue.
Conclusion
The flagella staining is a kind of motility test in the bacteria, which classifies them depending upon the presence, absence, number and arrangement of the flagella under the light microscope.
The content was so good to me