Urease test is an analytical method practised to identify the urease positive and negative organisms based on the production of cytosolic urease enzyme. It distinguishes the Proteus species from the non-lactose fermenting members belonging to the Enterobacteriaceae family.
Urease test makes the use of urea base. Conventionally, Stuart formulated urea broth was used to identify the rapid urease positive Proteus species. Later on, Christensen formulated urea agar to identify slow urease positive organisms belonging to the Enterobacteriaceae family.
Urease test comes into use to check whether the microorganism can produce “Urease” or not. It involves the splitting of urea into end products like alkaline ammonia and carbon dioxide by the addition of water.
This post discusses the definition, principle, procedure and test results of the urease test. You would also get to know the composition and preparation of the test media, along with a brief overview of the urease test.
Content: Urease Test
Definition
Urease test is a biochemical method that demarcates the difference between rapid positive, slow positive and negative urease test organisms based on the existence of the urease enzyme. Urease catalyses the hydrolysis of urea into ammonia and carbon dioxide. The urease test results are characterized by any colour change in the pH indicator (phenol red).
- The pH at 6.8 and between 7.5-8.1, the colour of the phenol red indicator appears yellow-orange and red, respectively.
- The colour of the phenol red turns fluorescent pink at a pH above 8.2.
Urea is an acidic compound, which on hydrolysis produces an alkaline by-product, namely ammonia. Therefore, the colour change of the pH indicator is due to the formation of ammonia (end product).
Overview
Before discussing the theory of the urease test, let us study some important terms that we must have an idea of.
Urease
It refers to urease aminohydrolase, which exists as a cytosolic enzyme that breaks urea into the by-products (carbon dioxide and ammonia). It functions as a nitrogen metabolizer, as it degrades the chief nitrogenous product “Urea”. Urease is a nickel-containing metalloenzyme.
It exists in archaea, bacteria, unicellular eukaryotes etc. and a member of the aminohydrolases and phosphodiesterases superfamily. Urease is dependent on the substrates like urea and hydroxyurea. Its reactivity may alter from species to species.
Urea
It refers to a chief nitrogenous end product formed by the decarboxylation of protein subunits (amino acids). Urea is a diamide of carbonic acid. It is a chemical compound that comprises two –NH2 groups connected via the carbonyl (C=O) functional group.
Urea Broth
A scientist named Stuart formulated urea broth, which is also known as Stuart’s Urea broth media.
- It was commonly used to get rapid results of the urease test, and the bacteria giving quick positive results are Proteus sp, Providencia sp etc.
- This media has a high buffering capacity and limited nutrients, due to which the organisms degrading urea slowly cannot be identified.
Urea Agar Media
A scientist named Christensen formulated urea agar in the year 1946, due to which the media is named after the name of the scientist. It contains a urea base and a pH indicator (phenol red).
The peptone contained in the media encourages faster growth and reaction time of the organisms. Dextrose contained in the media helps stimulate the urease activity in the microorganisms to make them efficient to hydrolyze urea.
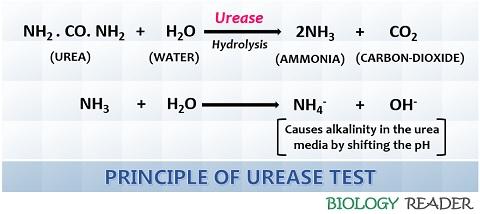
Principle of Urease Test
The mechanism of the urease test depends on the hydrolysis reaction catalyzed by the urease enzyme. Ureases split or hydrolyze the nitrogenous compound (urea) into the by-products like ammonia and carbon dioxide.
The organisms capable of hydrolyzing acidic urea (Proteus sp, Providencia sp etc.) can produce a sufficient amount of basic ammonia and a gaseous end-product carbon dioxide. The ammonia further protonates into ammonium and hydroxyl ions that cause alkalinity in the media.
Thus, the formation of bases results in changing the pH value of the pH indicator, which eventually changes the colour of the media. The pH shift of the media from acidic (6.8) to alkaline (> 8.2) changes the colour of the media from yellow-orange to pink-red.
Few organisms like Klebsiella and Enterobacter species can change the pH and the media’s colour but generally give delayed results by slowing metabolizing the urea. Oppositely, some microorganisms like Escherichia coli lacks a urease enzyme, due to which they are incapable of splitting urea.
Test Media
The urease test uses either Christensen’s urea agar medium or Stuart’s urea broth medium.
Stuart’s Urea Broth
The composition of Stuart’s urea broth medium requires the following constituents:
- Yeast extract: 0.1 g
- Potassium phosphate (monobasic): 9.1 g
- Potassium phosphate (dibasic): 9.5 g
- Urea: 20 g
- Distilled water: 1 L
Preparation: To prepare urea broth, accurately weigh the above ingredients of Stuart’s media and add 1000 ml of distilled water. Filter sterilize the solution with the filter paper having a pore size of 0.45 mm. After that, autoclave the broth medium at 121 degrees Celsius for 15-20 minutes.
Christensen’s Urea Agar
The composition of Christensen’s urea agar medium requires the following constituents:
- Urea: 20 g
- Monopotassium phosphate: 2 g
- Peptone: 1 g
- Dextrose: 1 g
- Phenol red: 0.012 g
- Agar: 15 g
- Distilled water: 1L
Preparation: To prepare a urea agar slant, accurately weigh the above six contents of Christensen’s media and add 100 ml of distilled water. Filter sterilize the urea-based solution and 100ml of distilled water through the filter paper having 0.45 mm porosity.
Take 20 grams of agar media into the sterile flask and autoclave it at 121 degrees Celsius for 15-20 minutes. Allow the agar media to stand for a while until it reaches 50-55 degrees Celsius. Afterwards, aseptically transfer the 100 ml of a solution containing urea base into it.
Thoroughly mix the contents by shaking the flask back and forth. Then, add 3-4 ml of the media into the sterile tubes and keep them in an inverted position until it solidifies. After solidification, store the test tubes at 4-8 degrees Celsius under refrigeration.
Urease Test Procedure
The protocol of the urease test includes the following steps:
Experiment using Stuart’s urea broth media:
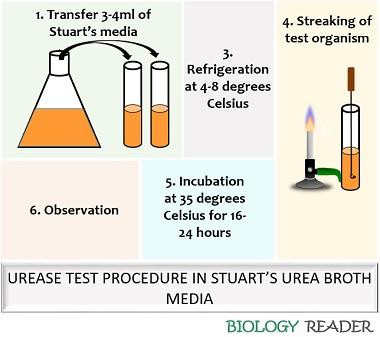
- After preparation and refrigerating the media, transfer the tubes into the laminar airflow.
- Dip the sterilized inoculating loop into Stuart’s urea broth medium.
- Incubate the culture tubes for 16-24 hours at 35 degrees Celsius.
- Observe the culture tubes for the appearance of a pink-red colour.
Experiment using Christensen’s urea agar media:
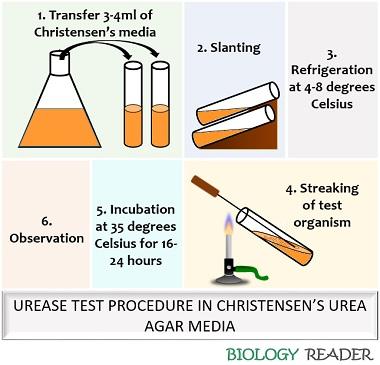
- After preparation and refrigerating the media, transfer the slants into the laminar airflow.
- Sterilize the inoculating loop over a flame and streak the test organism from the isolated colony over Christensen’s urea agar media.
- Incubate the culture tubes for at least 16 hours at 35 degrees Celsius.
- Regularly observe the culture tubes for any colour change, as few bacteria take time to split urea.
Test Results
The urease test results in Christensen’s urea agar and Stuart’s urea broth media can be concluded simultaneously by the given result interpretations.

Positive result: The whole media turns fluorescent pink-red colour within 16-24 hours.
Delayed positive result: The media slightly turns pink in colour and requires prolonged incubation of 6 days.
Negative result: The colour and the pH of the media will remain the same.