MacConkey agar media was first introduced by a scientist named Alfred Theodore MacConkey in the year 1890s. It is primarily exercised to isolate and differentiate the non-fastidious, gram-negative bacilli belonging to the Enterobacteriaceae family.
Previously, it was first considered as “Solid differential media”. But now, both liquid broth and solid culture MacConkey media preparations are generally practised in the laboratories for the selective as well as differential growth of microorganisms.
MacConkey agar media includes various constituents like bile salt, lactose and neutral red, which serve as the indicator system. It helps us to study and identify the growth pattern of the bacterial isolates based on the bile salt precipitation, lactose fermentation and formation of acid.
In this context, we will study the definition, composition, media preparation and principal role of the different constituents in the MacConkey agar media. Also, the result interpretations and the types of modified MacConkey agar media are explained along with the intended uses and limitations of the culture media.
Content: MacConkey Agar Media
Definition of MacConkey Agar Medium
MacConkey agar media serves as the indicator, selective and differential media, which determines the growth or presence of the gram-negative enteric bacilli from the different samples that are important for the sanitary purpose. It functions as a selective media, which ensures the growth of gram-negative enteric bacilli by restricting the growth of other organisms.
MacConkey agar media is also employed as a differential media by which one can easily discriminate the closely related gram-negative bacteria, depending upon the lactose fermentation and acid production. It also acts as the indicator media, which indicates the presence or absence of lactose fermenters by a definite colour change in the developing colonies as well as the media.
Appearance
- In dehydrated form: The media appears beige-pink and homogenous.
- In prepared form: The media appears slightly lustrous and pinkish-red in colour.
Composition of MacConkey Agar Media
Following are the ingredients to prepare 1L of the MacConkey agar media:
- Peptone: 7 g
- Proteose peptone: 3 g
- Lactose: 10 g
- Bile salts: 1.5 g
- Sodium chloride: 5 g
- Neutral red: 0.03 g
- Crystal violet: 0.001 g
- Agar: 13.5 g
- Distilled water: 1000 ml
Role of MacConkey Agar Media Components
The proteases and peptones provide vital elements and the sole N-source for the growth of gram-negative rods. Lactose is a fermentable carbohydrate that provides a sole C-source to promote bacterial growth.
Bile salt behaves as a selective agent that particularly suppresses the gram-positive organisms to grow. Gram-negative enteric bacteria are resistant to bile salts by having a bile-resistant outer membrane.
Sodium chloride functions to supply essential electrolytes for the cell growth of gram-negative organisms and also maintains the osmotic concentration.
Neutral red is a pH indicator dye, which allows us to discriminate organisms based on the colour change. When bacteria produce a sufficient amount of acids, the pH of the media falls, thereby resulting in the colour change of the neutral red pH indicator into pink.
Crystal violet is also a selective agent that mainly restricts the growth of gram-positive bacteria. Agar acts as a solidifying agent of the media.
Preparation of the Media
- Weigh all the contents accurately and dissolve them by adding 1L of sterile water into a sterilized conical flask.
- Then, put a cotton plug into the mouth of the conical flask to avoid any contamination.
- After that, mix the contents thoroughly over the hot plate by shaking the flask in a clockwise and anticlockwise direction.
- After getting a homogenous solution, subject the mixture to the autoclave sterilization for 30 minutes at 121 degrees Celsius, under 15lps pressure.
- Finally, take out the flask from the autoclave directly to the laminar airflow chamber for the plating, and the remaining media can be kept in a refrigerator for further use.
Principle
The MacConkey agar media principally help in the isolation and differentiation of the gram-negative bacterial isolates. The property of differentiating bacteria depends upon the capability of the organisms to ferment lactose. 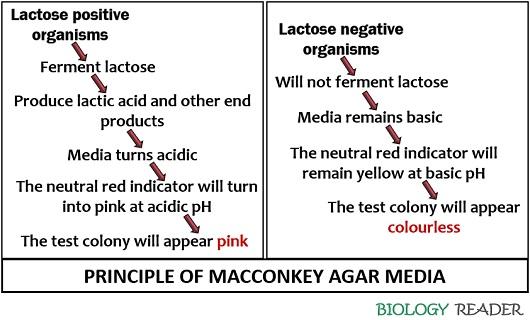
The lactose fermenting organisms produce a certain amount of acid due to fermentation and dropdown the pH of the media. As the pH drops below 6.8, the neutral red indicator will turn into pink that will be absorbed by the test organism.
Strong lactose fermenters will utilize lactose to form enough acids, resulting in the bile precipitation around the bacterial growth. The precipitation of bile salt gives a hazy appearance around the growth, and the colony appears as a pink halo.
In contrast, the weaker lactose fermenting bacteria will not form a pink halo, and the non-lactose fermenting bacteria will look colourless.
Result Interpretation
The growth and colour of the bacteria in the culture media have three possible outcomes.
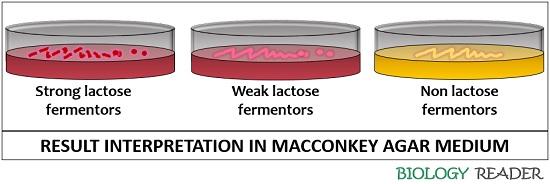
- Strong lactose fermenters: Their presence is indicated by the development of pink colonies encircled by pink halos (zones of precipitated bile salt). Strong lactose fermentors include Klebsiella species, Escherichia species etc.
- Weak lactose fermenters: Their presence is visualized by the growth of pink colonies after 48 hours of the incubation period. They do not form a zone of precipitated bile. Weak lactose fermenters include Citrobacter species, Serratia species etc.
- Non-lactose fermentors: Their presence is characterized by the growth of colourless colonies over the transparent MacConkey agar surface. Non-lactose fermentors include Salmonella species, Proteus species etc.
Modifications in the MacConkey Agar Media
- MacConkey media without CV: The MacConkey agar media without crystal violet is less selective as it allows the development of Staphylococcus and Enterococcus species. It also helps in determining the Mycobacterium fortuitum and M. chelonae from other fast-growing Mycobacteria.
- Controlled swarming MacConkey agar media: It lacks either crystal violet or salt in order to control the flocking of Proteus species.
- Sorbitol MacConkey agar media: It is an alternative to MacConkey agar media that uses sorbitol as a sole carbohydrate source in place of lactose.
- MacConkey broth: It works as a selective enrichment broth, using which the subculturing of the test organisms can be practised.
Intended Use
MacConkey agar works as a selective medium that isolates the gram-negative microorganisms from different sources like urine, stool, food samples etc. It also behaves as a differential media that discriminates between the closely related gram-negative enteric bacilli.
Limitations
MacConkey agar medium has a drawback that it gives presumptive test results of the isolated organisms. For the confirmatory results, the test organism needs further subculturing and biochemical tests. There is another limitation where some strains of the Proteus species may flock on this medium.